IB Chemistry Review
IB Chemistry notes with SL and HL topics, including the Drugs and Medicines and the Environmental Chemistry options.
Stats
Alexa Rank:
Popular in Country:
Country Alexa Rank:
language: en
Response Time: 0.192046
SSL: Enable
Status: up
Code To Txt Ratio
Word Count 134
Links
ratio 4.1867517595673
SSL Details
SSL Issuer:
Issuer: GTS CA 1C3
Valid From: 2022-06-06 08:59:22
Expiration Date: 2022-08-29 08:59:21
SSL Organization:
Signature 09ecf5a37f8210716bbf4aa859e8fcd17ca0d09f
Algorithm: RSA-SHA256
Found 13 Top Alternative to Ibchemistry-review.blogspot.com



 Ibalchemy.com
Ibalchemy.com
IB Alchemy – IB Chem Notes for New 2016 Syllabus
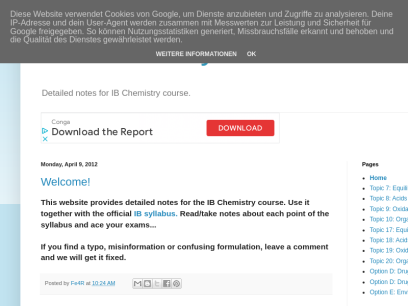
Home Standard Level 1. Stoichiometric relationships 1.1 Introduction to the particulate nature of matter and chemical change 1.2 The mole concept 1.3 Reacting masses and volumes 2. Atomic structure 2.1 The nuclear atom 2.2 Electron configuration 3. Periodicity 3.1 Periodic table 3.2 Periodic trends 4. Chemical bonding and structure 4.1...
 Wordpress.com
Wordpress.com
WordPress.com: Fast, Secure Managed WordPress Hosting
Create a free website or build a blog with ease on WordPress.com. Dozens of free, customizable, mobile-ready designs and themes. Free hosting and support.
 Myibchemistry.blogspot.com
Myibchemistry.blogspot.com
IB Chemistry
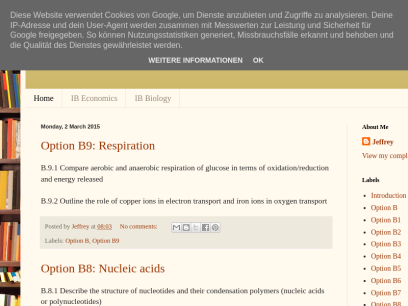
Option B9: Respiration B.9.1 Compare aerobic and anaerobic respiration of glucose in terms of oxidation/reduction and energy released B.9.2 Outline the role of copper ions in electron transport and iron ions in oxygen transport Posted by Jeffrey at 08:03 No comments: Email ThisBlogThis!Share to TwitterShare to FacebookShare to Pinterest Labels:...
 Mrwengibchemistry.com
Mrwengibchemistry.com
IB Chemistry Mr Weng
The most comprehensive set of IB Chemistry resources available online. Past paper online quizzes for all topics 1-11 (21) SL and HL. Tutorial videos, Internal Assessment (IAs and EEs) advice for topics and questions. Exam preparation course and a lot more! No other site has the depth and breath for IB Chemistry.

 Sciencesfp.com
Sciencesfp.com
San Francisco de Paula, Science Department. - Department Home page
Science Department, Colegio Internacional de Sevilla, San Francisco de Paula
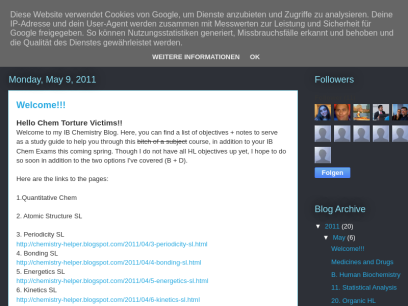
 Chemistry-helper.blogspot.com
Chemistry-helper.blogspot.com
IB Chem Helper
I. Energy Calculate the energy value of food from enthalpy of combustion data Use the equation: E = mcΔT. E = Energy M= Mass of the sample heated (usually water, NOT the product being burned). C= Specific heat capacity (water = 4.18 J g-1 C-1) ΔT = Change in temperature...

 Weebly.com
Weebly.com
Free Website Builder: Build a Free Website or Online Store | Weebly
Weebly’s free website builder makes it easy to create a website, blog, or online store. Find customizable templates, domains, and easy-to-use tools for any type of business website.
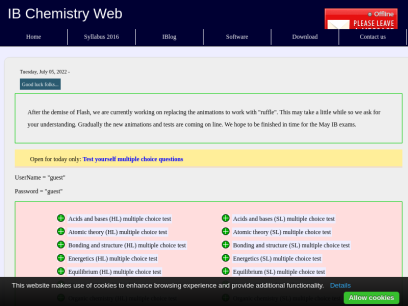
 Ibchem.com
Ibchem.com
IB Chemistry revision notes and syllabus
Please sign me up to receive the latest information and news about IB chemistry and the other Group IV sciences Name Email Country Select a country or territory Afghanistan Albania Algeria American Samoa Andorra Angola Anguilla Antarctica Antigua and Barbuda Argentina Armenia Aruba Ashmore and...
Technologies Used by ibchemistry-review.blogspot.com
Dns Records of ibchemistry-review.blogspot.com
A Record: 142.250.185.97AAAA Record: 2a00:1450:4001:80f::2001
CNAME Record: ibchemistry-review.blogspot.com
NS Record:
SOA Record:
MX Record:
SRV Record:
TXT Record:
DNSKEY Record:
CAA Record:
 199.34.228.72
199.34.228.72
 United States
United States
 Valid SSL
Valid SSL
